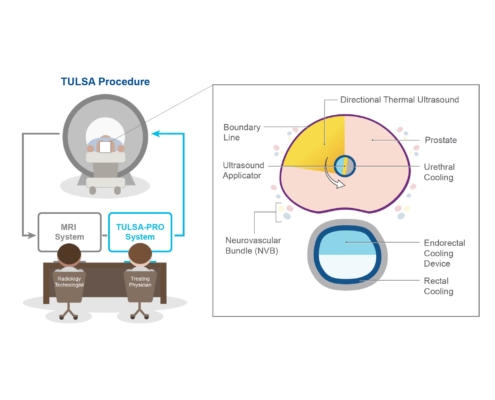
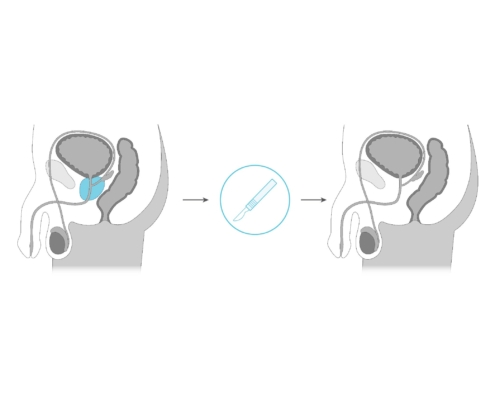
What is the TULSA Procedure?
The Transurethral Ultrasound Ablation (TULSA) Procedure uses directional thermal ultrasound to ablate (destroy) diseased prostate tissue. It has been demonstrated to be safe and effective at ablating prostate tissue in men with localized, intermediate-risk prostate cancer. The TULSA-PRO® is FDA cleared for use in the United States, Canada and Europe.
What is Radical Prostatectomy?
Radical Prostatectomy is a procedure performed using robotic technique or open surgery that involves the removal of your prostate and seminal vesicles (and sometimes nearby lymph nodes). For men with localized, intermediate-risk prostate cancer, RP is recommended to be a standard-of-care treatment and has been proven in large randomized controlled trials to reduce the long-term risk of dying from prostate cancer.
Participant Journey Map
Participating Centers
Please click on the study center image and scroll to the bottom of the page for participating center contact information.